 
The formula weight is simply the weight in atomic mass units of all the atoms in a given formula. When calculating molecular weight of a chemical compound, it tells us how many grams are in one mole of that substance. These relative weights computed from the chemical equation are sometimes called equation weights.įinding molar mass starts with units of grams per mole (g/mol). For bulk stoichiometric calculations, we are usually determining molar mass, which may also be called standard atomic weight or average atomic mass.įormula weights are especially useful in determining the relative weights of reagents and products in a chemical reaction. This is not the same as molecular mass, which is the mass of a single molecule of well-defined isotopes. This is how to calculate molar mass (average molecular weight), which is based on isotropically weighted averages. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The percentage by weight of any atom or group of atoms in a compound can be computed by dividing the total weight of the atom (or group of atoms) in the formula by the formula weight and multiplying by 100. If the formula used in calculating molar mass is the molecular formula, the formula weight computed is the molecular weight. This site explains how to find molar mass. The reason is that the molar mass of the substance affects the conversion. To complete this calculation, you have to know what substance you are trying to convert. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance.Ī common request on this site is to convert grams to moles. 109535).In chemistry, the formula weight is a quantity computed by multiplying the atomic weight (in atomic mass units) of each element in a chemical formula by the number of atoms of that element present in the formula, then adding all of these products together. Before placing in container D, check the pH with pH Universal indicator strips (Cat.No. To neutralize alkyl sulfates, add dropwise (from a dropping funnel) to concentrated ice-cool Ammonia solution (Cat.No. Alkyl sulfates are carcinogenic Take particular care to avoid inhalation and skin contact. Keep container tightly closed.ĩ Carcinogenic compounds and flammable compounds labelled "Highly toxic" or "Toxic": container F. Average atomic mass of carbon is 12.011 what is the abundance of C12 in nature 14624066. 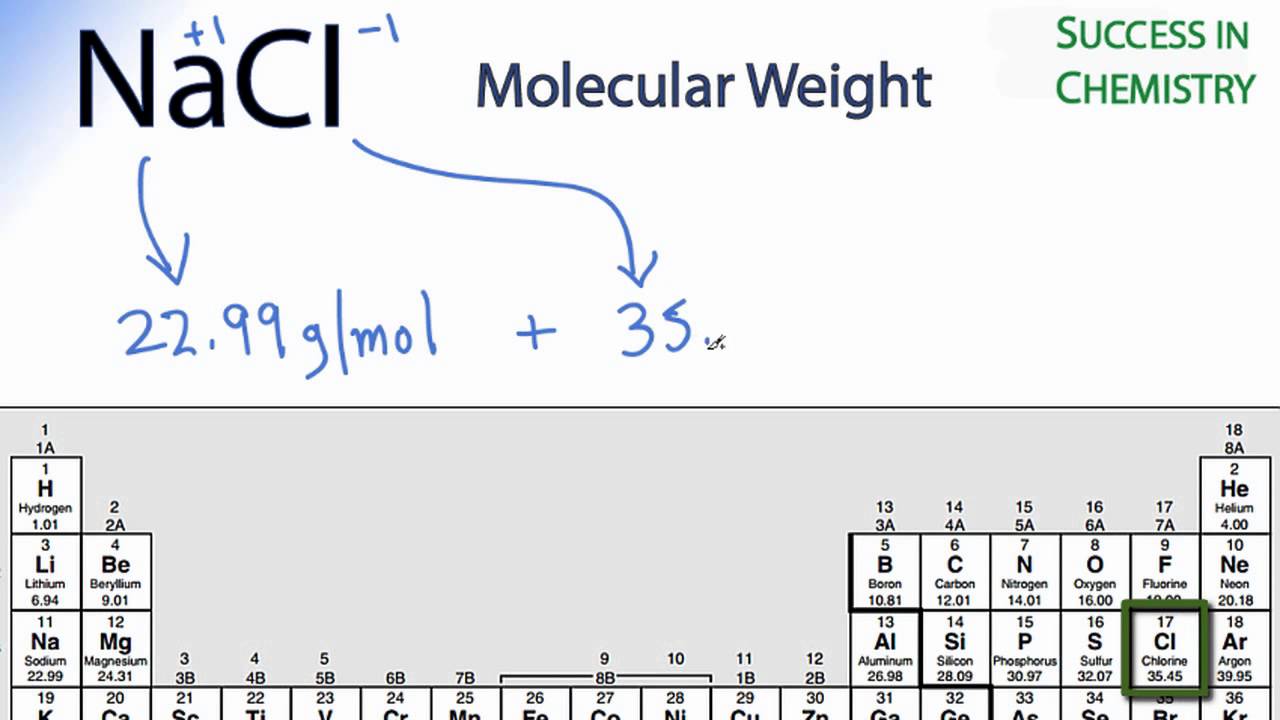
It is also a naturally occurring isotope, meaning that it is not produced in a laboratory. P403 + P233: Store in a well-ventilated place. Carbon-13: Carbon-13 is one of only two stable isotopes of carbon the other being carbon-12. P308 + P313: IF exposed or concerned: Get medical advice/ attention. Remove contact lenses, if present and easy to do. P305 + P351 + P338: IF IN EYES: Rinse cautiously with water for several minutes. It is also a naturally occurring isotope. Call a POISON CENTER/doctor if you feel unwell. What is the molar mass of carbon-13 Carbon-13: Carbon-13 is one of only two stable isotopes of carbon the other being carbon-12. P304 + P340 + P312: IF INHALED: Remove person to fresh air and keep comfortable for breathing. P303 + P361 + P353: IF ON SKIN (or hair): Take off immediately all contaminated clothing. 
P210: Keep away from heat, hot surfaces, sparks, open flames and other ignition sources. P201: Obtain special instructions before use. H372: Causes damage to organs through prolonged or repeated exposure. H225: Highly flammable liquid and vapour. Ph Eur and ACS.Ĭarbon disulfide for spectroscopy Uvasol®. Furthermore the transmittance is specified in accordance with Reag. In all specifications the minimum transmittance for 5 typical wavelengths are identified. Uvasol® solvents offer best UV transmittance. The refinement process allows a greater degree of security in applications and avoids misinterpretation of analytical results caused by traces of UV, IR and fluorescence contamination. The Uvasol® solvents range has been specially designed for spectroscopy and other applications requiring solvents of the highest spectral purity. Accurate analytic results in UV/VIS and infrared spectroscopy depend on the use of very pure solvents for sample preparation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
